 However the conformation of the second extracellular loop is entirely different between the two structures. 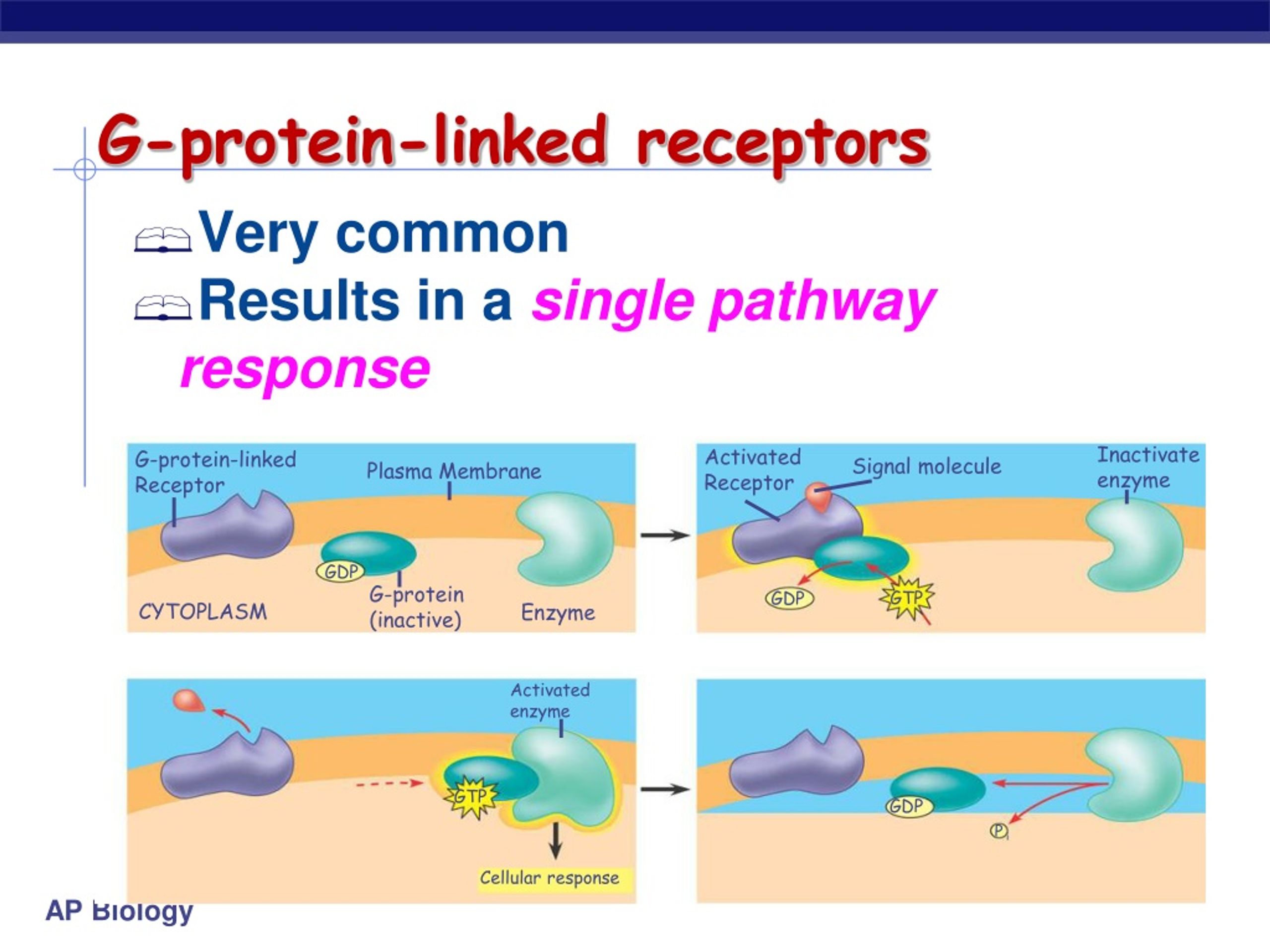
This human β 2-adrenergic receptor GPCR structure, proved to be highly similar to the bovine rhodopsin in terms of the relative orientation of the seven transmembrane helices. This was followed immediately by a higher resolution structure of the same receptor ( 2RH1). In 2007, the first structure of a human GPCR was solved ( 2R4R, 2R4S). While the main feature, the seven transmembrane helices, is conserved, the relative orientation of the helices differ significantly from that of bacteriorhodopsin. In 2000, the first crystal structure of a mammalian GPCR, that of bovine rhodopsin ( 1F88), was solved. Some seven transmembrane helix proteins (such as channelrhodopsin) that resemble GPCRs may contain different functional groups, such as entire ion channels, within their protein.Įarly structural models for GPCRs were based on their weak analogy to bacteriorhodopsin for which a structure had been determined by both electron diffraction ( PDB 2BRD, 1AT9) and X ray-based crystallography ( 1AP9). These extracellular loops also contain two highly-conserved cysteine residues that form disulfide bonds to stabilize the receptor structure. The extracellular parts of the receptor can be glycosylated. GPCRs are integral membrane proteins that possess seven membrane-spanning domains or transmembrane helices (Figure 1).
0 Comments
Leave a Reply. |



 RSS Feed
RSS Feed
